Parkinson's disease uncovers an underlying sensitivity of subthalamic nucleus neurons to beta-frequency cortical input in vivo.
It is unclear why people with Parkinson's disease have exaggerated rhythmic electrical activity at so-called beta frequency across many brain areas. Using recordings from patients undergoing neurosurgery and a rodent model of the disease, we show that the electrical impulses of single nerve cells in one of these brain areas, the subthalamic nucleus, are more sensitive to beta-frequency input than other rhythms.
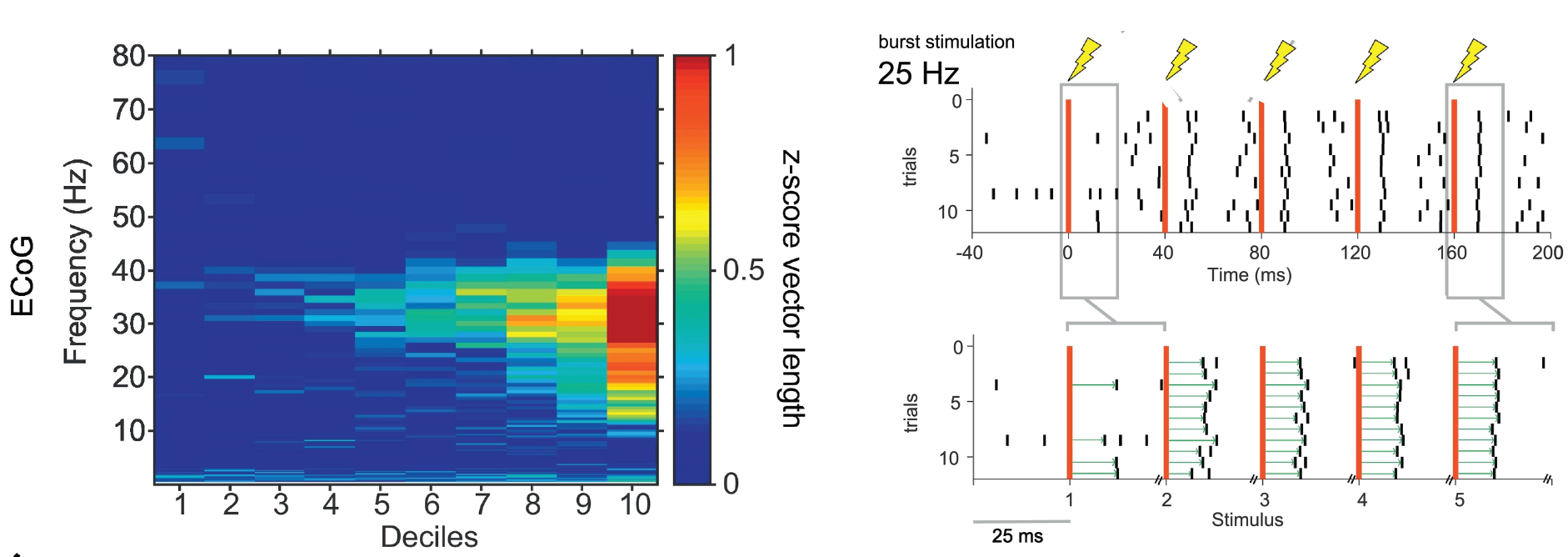
Abnormally sustained beta-frequency synchronisation between the motor cortex and subthalamic nucleus (STN) is associated with motor symptoms in Parkinson's disease (PD). It is currently unclear whether STN neurons have a preference for beta-frequency input (12-35 Hz), rather than cortical input at other frequencies, and how such a preference would arise following dopamine depletion. To address this question, we combined analysis of cortical and STN recordings from awake human PD patients undergoing deep brain stimulation surgery with recordings of identified STN neurons in anaesthetised rats. In these patients, we demonstrate that a subset of putative STN neurons is strongly and selectively sensitive to magnitude fluctuations of cortical beta oscillations over time, linearly increasing their phase-locking strength with respect to the full range of instantaneous amplitude in the beta-frequency range. In rats, we probed the frequency response of STN neurons in the cortico-basal-ganglia-network more precisely, by recording spikes evoked by short bursts of cortical stimulation with variable frequency (4-40 Hz) and constant amplitude. In both healthy and dopamine-depleted rats, only beta-frequency stimulation led to a progressive reduction in the variability of spike timing through the stimulation train. This suggests, that the interval of beta-frequency input provides an optimal window for eliciting the next spike with high fidelity. We hypothesize, that abnormal activation of the indirect pathway, via dopamine depletion and/or cortical stimulation, could trigger an underlying sensitivity of the STN microcircuit to beta-frequency input.
2024. Neurobiol Dis, 201:106652.
2021. Nat Commun, 12(1):5185.
2022. NPJ Parkinsons Dis, 8(1):88.