A functional micro-electrode mapping of ventral thalamus in essential tremor.
A part of the brain called the ventral thalamus is often targeted for the surgical treatment of tremor. Here, we show that the targeted thalamus contains intermixed clusters of different nerve cells; some cells receive input from tremulous muscles whereas others seems to drive these muscles. This complex interplay of nerve cells suggests that tremor may be self-reinforcing.
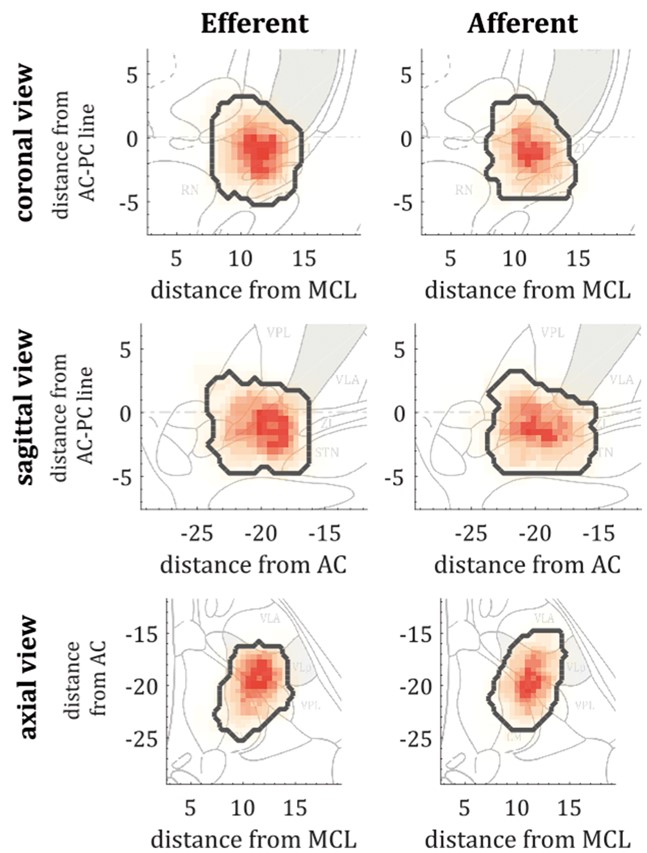
Deep brain stimulation enables the delivery of therapeutic interventions to otherwise inaccessible areas of the brain while, at the same time, offering the unique opportunity to record from these same regions in awake patients. The posterior ventrolateral thalamus has become a reliable deep brain stimulation target for medically-refractory patients suffering from essential tremor. However, the contribution of the thalamus in essential tremor, and even whether posterior ventrolateral thalamus is the optimal target, remains a matter of ongoing debate. There are several lines of evidence supporting clusters of activity within the posterior ventrolateral thalamus that are important for tremor emergence. In this study we sought to map the functional properties of these clusters through microelectrode recordings during deep brain stimulation surgery. Data were obtained from 10 severely affected patients with essential tremor (12 hemispheres) undergoing deep brain stimulation surgery. Our results demonstrate power and coherence maxima located in the inferior posterior ventrolateral thalamus and immediate ventral region. Moreover, we identified distinct yet overlapping clusters of predominantly efferent (driving) and afferent (feedback) activity, with a preference for more efferent contributors, consistent with a net role in the driving of tremor output. Finally, we demonstrate that resolvable thalamic spiking activity directly relates to background activity and that the strength of tremor may be dictated by phase relationships between efferent and afferent pockets in the posterior ventrolateral thalamus. Taken together, these results provide important evidence for the role of the inferior posterior ventrolateral thalamus and its border region in essential tremor pathophysiology. Such results progress our mechanistic understanding and promote the adoption of next-generation therapies such as high resolution segregated deep brain stimulation electrodes.
2025. Brain, 148(6):2093-2107.
2021. Brain, 144(3):781-788.
2025. Mov Disord, 40(9):1977-1982.