Cross-frequency coupling between gamma oscillations and deep brain stimulation frequency in Parkinson's disease.
Therapeutic deep brain stimulation in people with Parkinson’s is thought to suppress abnormal waves of nerve cell activity called beta oscillations in the brain system supporting voluntary movements. Here, we confirm this suppression and also show that the same stimulation promotes faster waves called gamma oscillations in the same brain system.
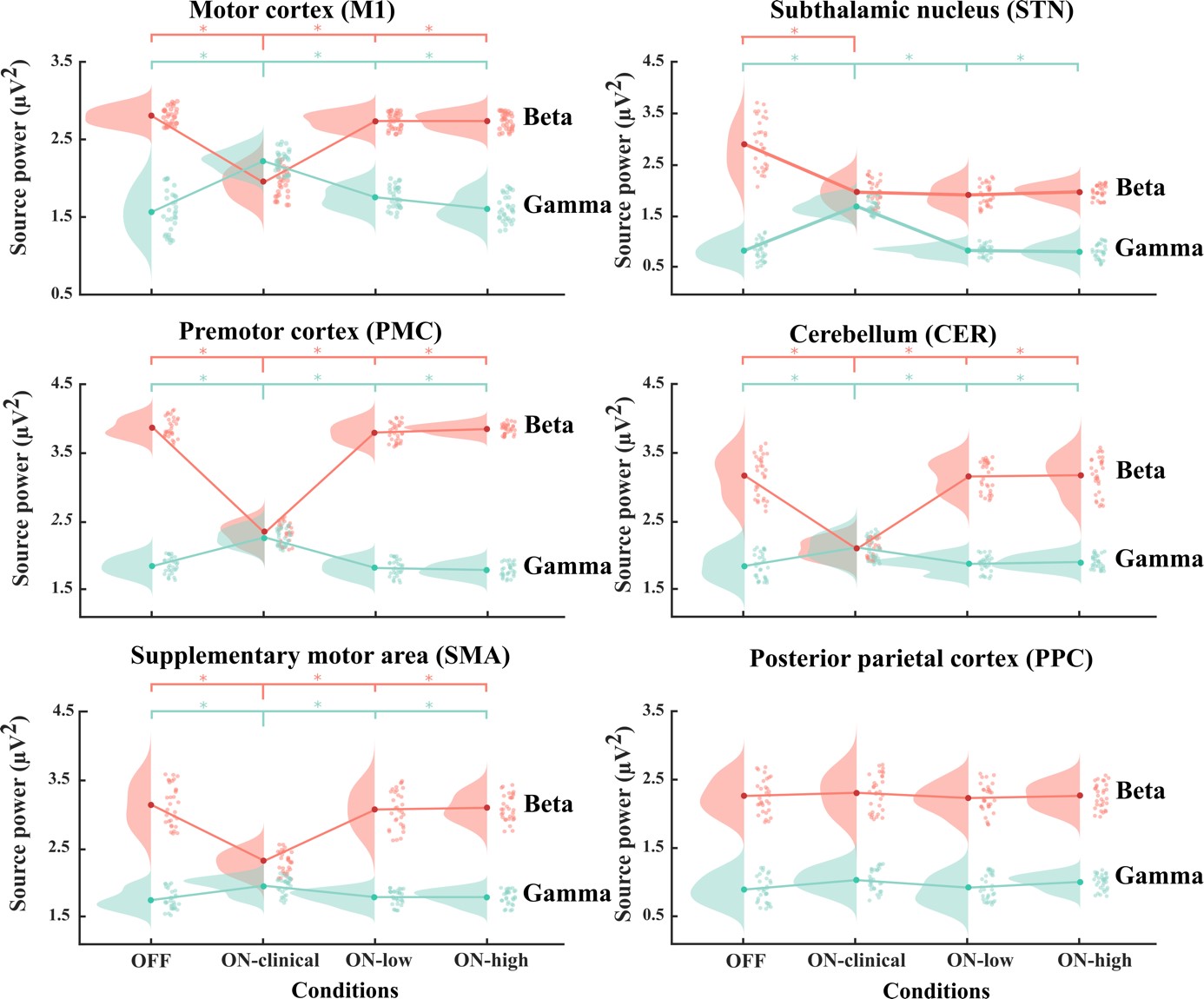
The disruption of pathologically enhanced beta oscillations is considered one of the key mechanisms mediating the clinical effects of deep brain stimulation on motor symptoms in Parkinson's disease. However, a specific modulation of other distinct physiological or pathological oscillatory activities could also play an important role in symptom control and motor function recovery during deep brain stimulation. Finely tuned gamma oscillations have been suggested to be prokinetic in nature, facilitating the preferential processing of physiological neural activity. In this study, we postulate that clinically effective high-frequency stimulation of the subthalamic nucleus imposes cross-frequency interactions with gamma oscillations in a cortico-subcortical network of interconnected regions and normalizes the balance between beta and gamma oscillations. To this end we acquired resting state high-density (256 channels) EEG from 31 patients with Parkinson's disease who underwent deep brain stimulation to compare spectral power and power-to-power cross-frequency coupling using a beamformer algorithm for coherent sources. To show that modulations exclusively relate to stimulation frequencies that alleviate motor symptoms, two clinically ineffective frequencies were tested as control conditions. We observed a robust reduction of beta and increase of gamma power, attested in the regions of a cortical (motor cortex, supplementary motor area, premotor cortex) and subcortical network (subthalamic nucleus and cerebellum). Additionally, we found a clear cross-frequency coupling of narrowband gamma frequencies to the stimulation frequency in all of these nodes, which negatively correlated with motor impairment. No such dynamics were revealed within the control posterior parietal cortex region. Furthermore, deep brain stimulation at clinically ineffective frequencies did not alter the source power spectra or cross-frequency coupling in any region. These findings demonstrate that clinically effective deep brain stimulation of the subthalamic nucleus differentially modifies different oscillatory activities in a widespread network of cortical and subcortical regions. Particularly the cross-frequency interactions between finely tuned gamma oscillations and the stimulation frequency may suggest an entrainment mechanism that could promote dynamic neural processing underlying motor symptom alleviation.
2021. Neurobiol Dis, 152:105287.
2023. Brain, 146(12):5015-5030.
2022. Brain, 145(1):237-250.