Association of specific biotypes in patients with Parkinson disease and disease progression.
To advise people affected by Parkinson’s about the future, and develop new and better treatments, we need to be able to reliably predict how Parkinson’s progresses. Here, with colleagues in China, we show that the pattern of brain volume identified on scanning is able to separate patient groups into those with slow or rapid progression in symptoms.
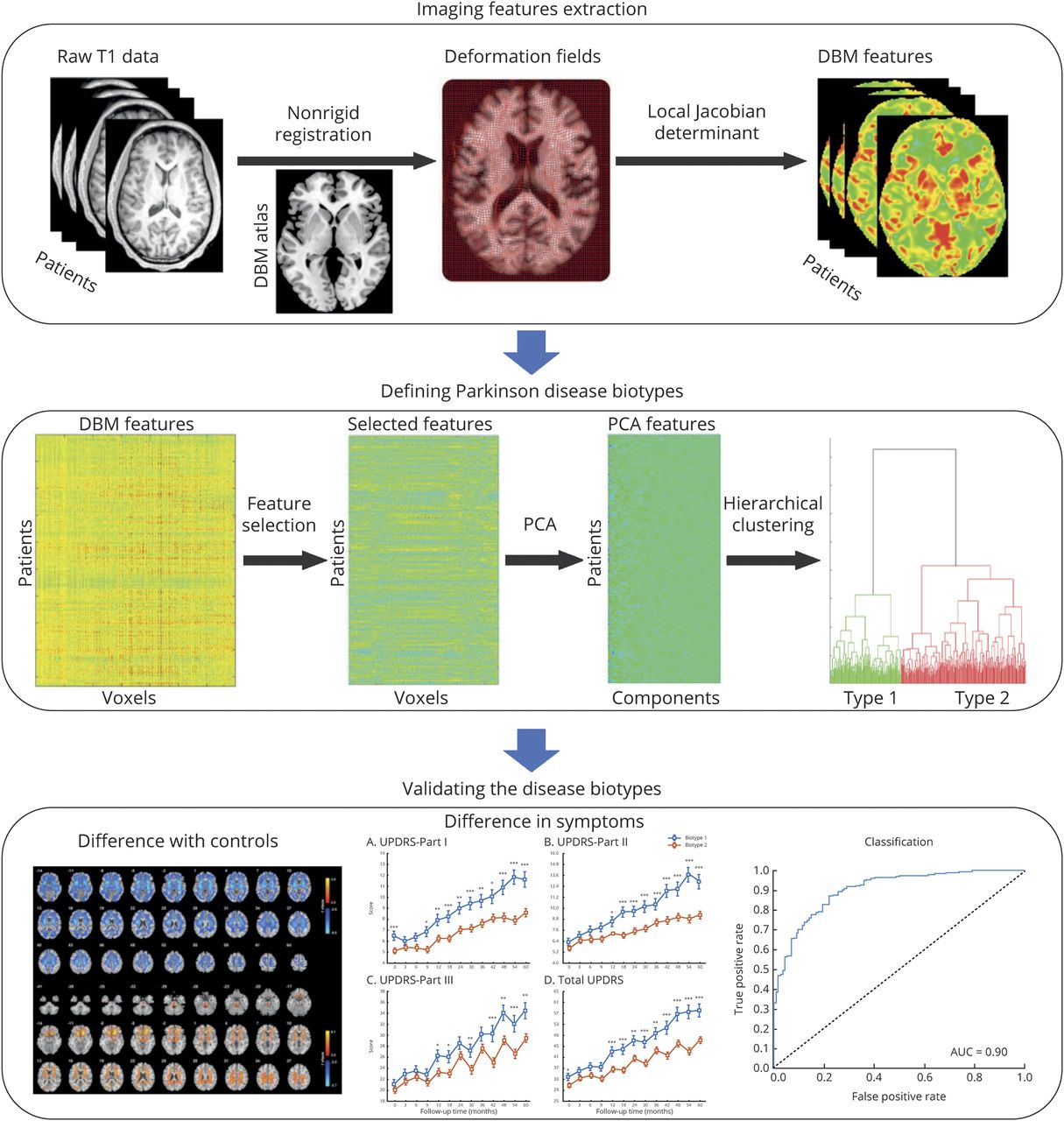
Objective: To identify biotypes in patients with newly diagnosed Parkinson disease (PD) and to test whether these biotypes could explain interindividual differences in longitudinal progression.
Methods: In this longitudinal analysis, we use a data-driven approach clustering PD patients from the Parkinson's Progression Markers Initiative (n = 314, age 61.0 ± 9.5, years 34.1% female, 5 years of follow-up). Voxel-level neuroanatomic features were estimated with deformation-based morphometry (DBM) of T1-weighted MRI. Voxels with deformation values that were significantly correlated (p < 0.01) with clinical scores (Movement Disorder Society-sponsored revision of the Unified Parkinson's Disease Rating Scale Parts I-III and total score, tremor score, and postural instability and gait difficulty score) at baseline were selected. Then, these neuroanatomic features were subjected to hierarchical cluster analysis. Changes in the longitudinal progression and neuroanatomic pattern were compared between different biotypes.
Results: Two neuroanatomic biotypes were identified: biotype 1 (n = 114) with subcortical brain volumes smaller than heathy controls and biotype 2 (n = 200) with subcortical brain volumes larger than heathy controls. Biotype 1 had more severe motor impairment, autonomic dysfunction, and much worse REM sleep behavior disorder than biotype 2 at baseline. Although disease durations at the initial visit and follow-up were similar between biotypes, patients with PD with smaller subcortical brain volume had poorer prognosis, with more rapid decline in several clinical domains and in dopamine functional neuroimaging over an average of 5 years.
Conclusion: Robust neuroanatomic biotypes exist in PD with distinct clinical and neuroanatomic patterns. These biotypes can be detected at diagnosis and predict the course of longitudinal progression, which should benefit trial design and evaluation.
2022. NPJ Parkinsons Dis, 8(1):88.
2020. J. Neurosci., 40(7):1571-1580.
