Secretagogin expression delineates functionally-specialized populations of striatal parvalbumin-containing interneurons.
The striatum is a brain area important for learning and making successful actions. Nerve cells called interneurons help control communication within striatum. We studied the molecular building blocks, connections and electrical activity of nerve cells in striatum, and show that two special types of interneuron could play different but complementary roles in controlling other cells and behaviour.
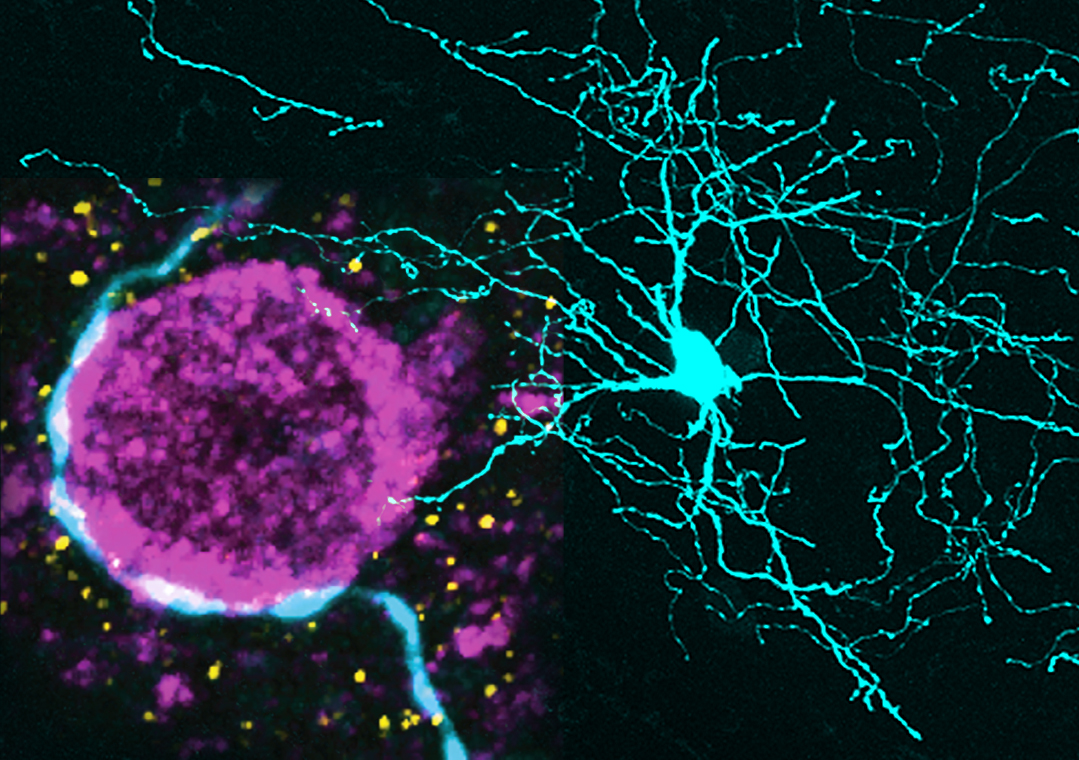
Corticostriatal afferents can engage parvalbumin-expressing (PV+) interneurons to rapidly curtail the activity of striatal projection neurons (SPNs), thus shaping striatal output. Schemes of basal ganglia circuit dynamics generally consider striatal PV+ interneurons to be homogenous, despite considerable heterogeneity in both form and function. We demonstrate that the selective co-expression of another calcium-binding protein, secretagogin (Scgn), separates PV+ interneurons in rat and primate striatum into two topographically-, physiologically- and structurally-distinct cell populations. In rats, these two interneuron populations differed in their firing rates, patterns and relationships with cortical oscillations in vivo. Moreover, the axons of identified PV+/Scgn+ interneurons preferentially targeted the somata of SPNs of the so-called 'direct pathway', whereas PV+/Scgn- interneurons preferentially targeted 'indirect pathway' SPNs. These two populations of interneurons could therefore provide a substrate through which either of the striatal output pathways can be rapidly and selectively inhibited to subsequently mediate the expression of behavioral routines.
2023. eNeuro, 10(7).