Dr Kouichi C. Nakamura

Dr Kouichi C. Nakamura
Dr. Kouichi Nakamura graduated in biological sciences at Kyoto University, Japan, in 2000. He then studied postnatal development of glutamatergic neuronal circuits under the supervision of Professor Takeshi Kaneko, and obtained his Ph.D. from the Graduate School of Medicine at Kyoto University in 2006. Dr. Nakamura then stayed in the Kaneko laboratory as a postdoctoral researcher to extend his studies on the postnatal development of the basal ganglia circuits. Dr. Nakamura joined the Magill Group as a Long-Term Fellow of the Human Frontier Science Program Organization in September 2009. After the completion of his Fellowship in September 2012, he continued his studies of the motor thalamus in the Unit as an MRC Investigator Scientist. Dr. Nakamura returned to Kyoto University in September 2013 to serve as an Assistant Professor, before rejoining the Magill Group in May 2016.
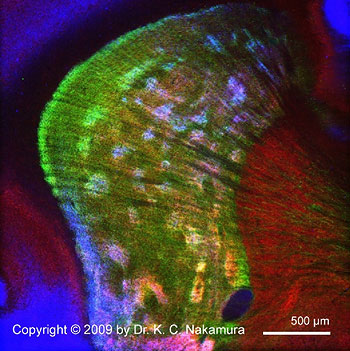
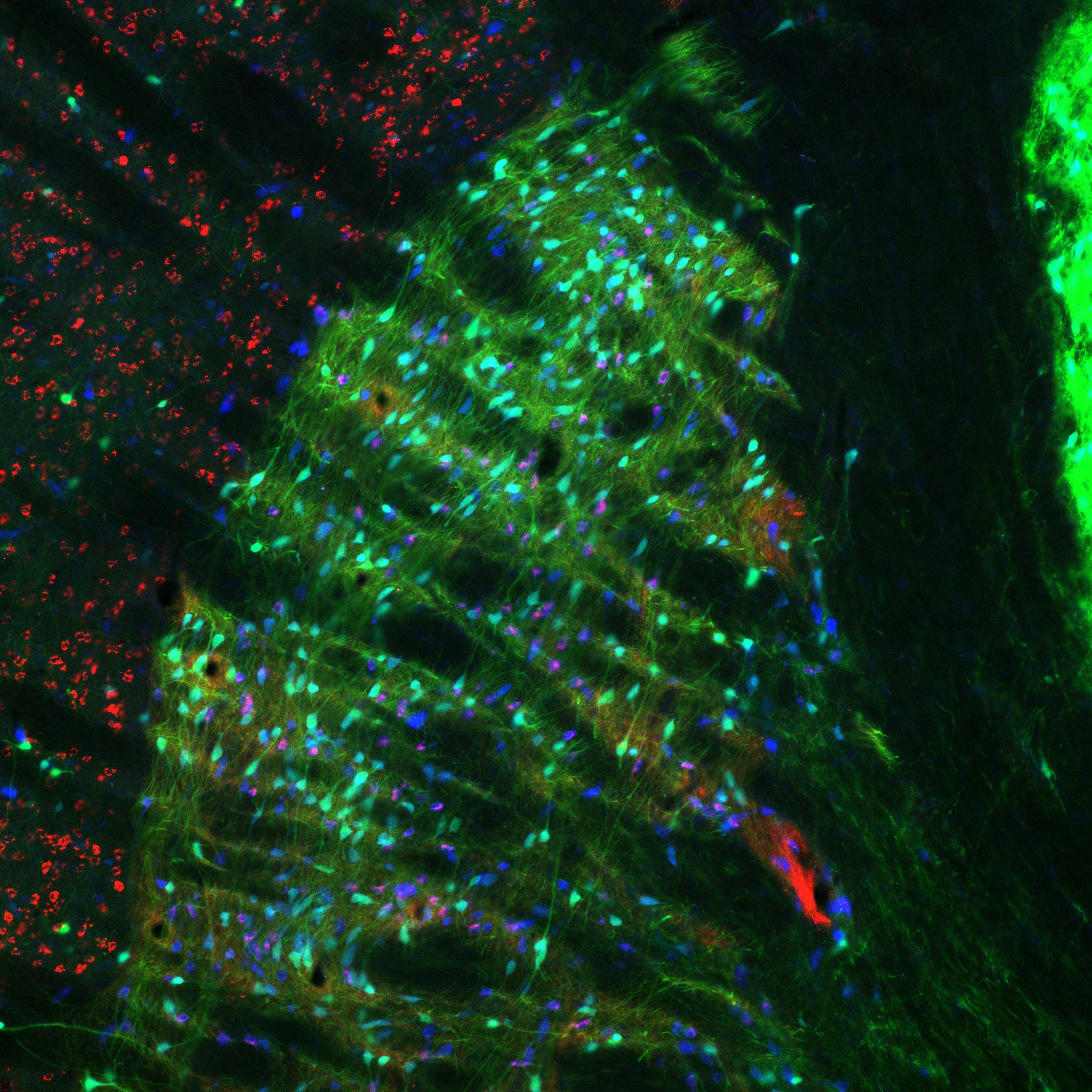
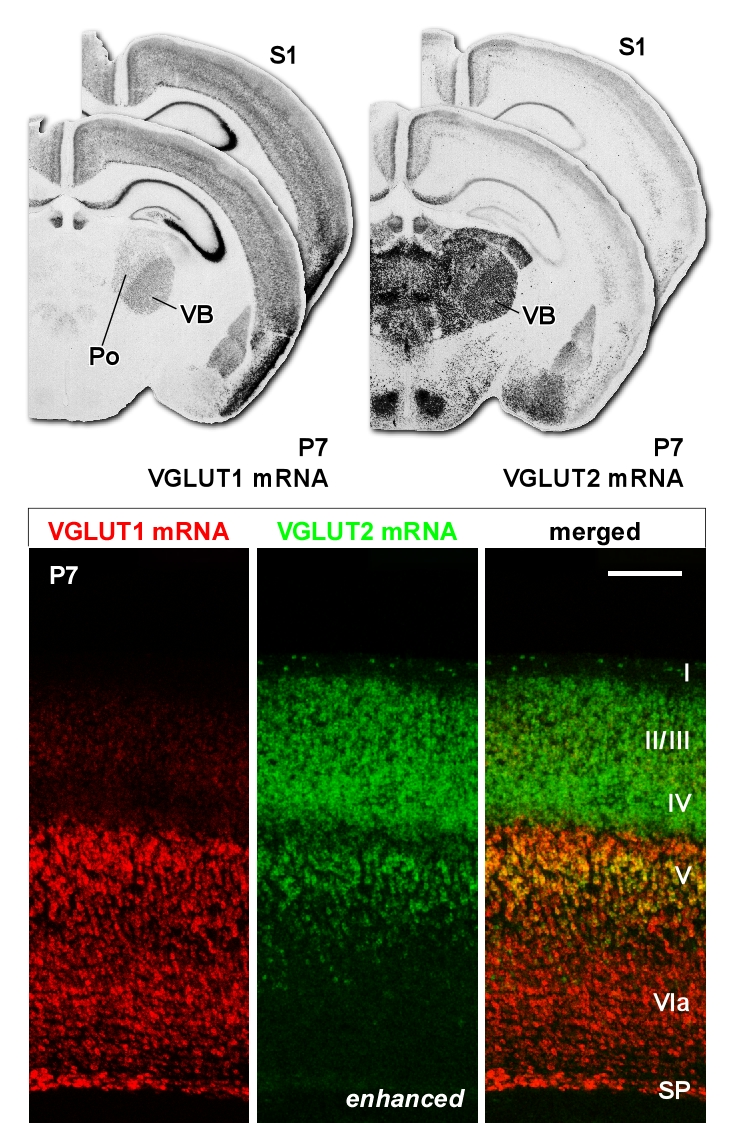
At the Unit, Dr. Nakamura used in vivo electrophysiological recording techniques, anatomical methods, and genetics-based approaches for monitoring and manipulating specified cell types to elucidate the principles governing neuronal communication within the basal ganglia and partner thalamic circuits.
Dr. Nakamura worked as a Senior Postdoctoral Neuroscientist in the Magill Group until March 2024. He continues to collaborate closely with the Group.
- Processing of movement-related information by neurons in the thalamus, basal ganglia, cerebral cortex and cerebellum.
- Behavioural correlates of neuronal information processing.
- Experimental models of Parkinson's disease.
- Develpment of CHAMBER (CHemoarchitectonic Atlas of the Mouse thalamus as a BNDU OpEn Resource) to provide publicly available anatomical reference for the delineation of brain structures, such as thalamic nuclei.