Distinct developmental origins manifest in the specialized encoding of movement by adult neurons of the external globus pallidus.
Understanding how nerve cells are born and develop helps clarify cell function in later life. Here, we show that two types of nerve cell in the external pallidum, a brain region important for movement, have different embryonic origins and molecular building blocks. These differences carry over to adulthood, so that the two cell types reflect movement with distinct patterns of electrical impulses.
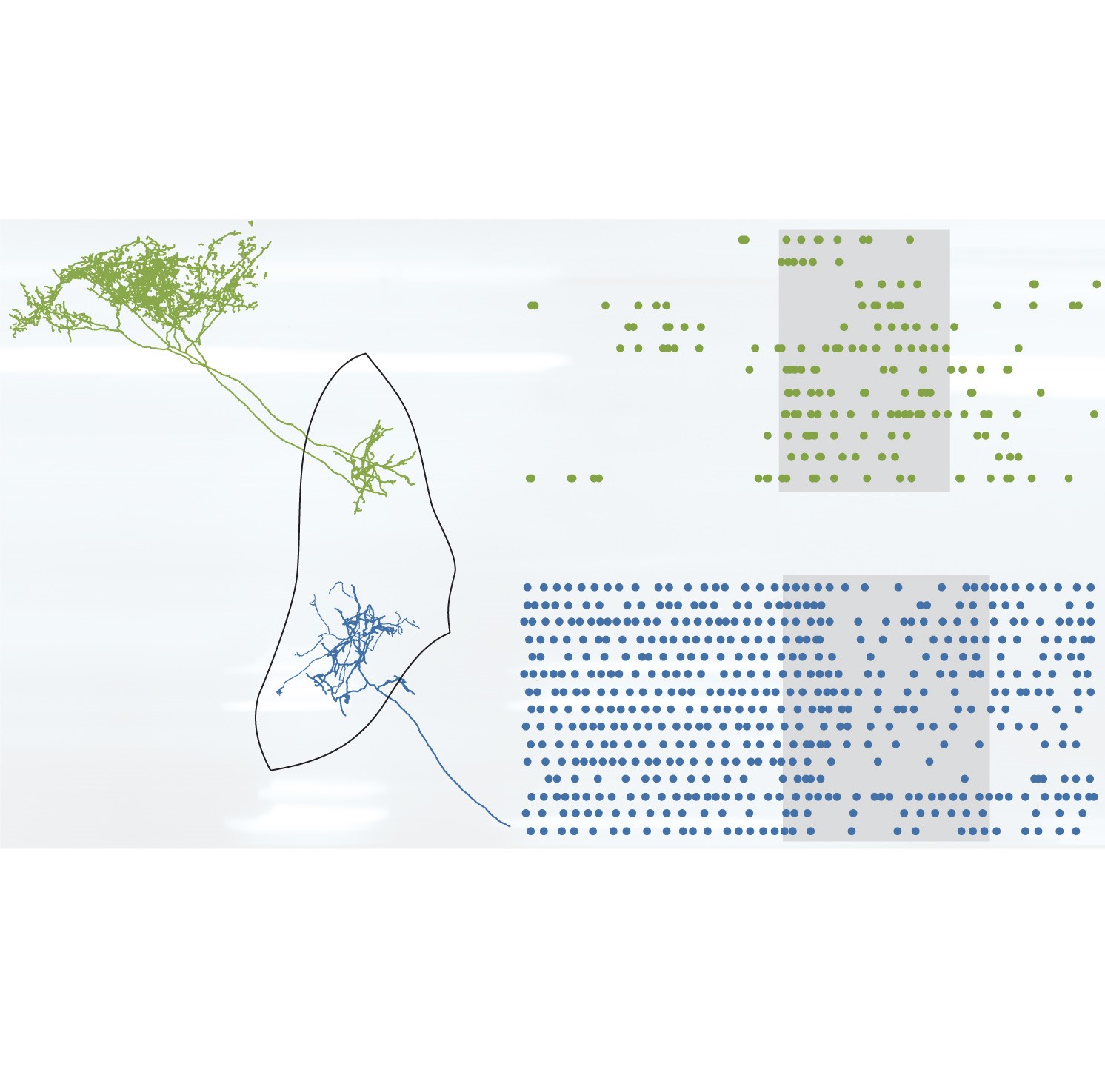
Transcriptional codes initiated during brain development are ultimately realized in adulthood as distinct cell types performing specialized roles in behavior. Focusing on the mouse external globus pallidus (GPe), we demonstrate that the potential contributions of two GABAergic GPe cell types to voluntary action are fated from early life to be distinct. Prototypic GPe neurons derive from the medial ganglionic eminence of the embryonic subpallium and express the transcription factor Nkx2-1. These neurons fire at high rates during alert rest, and encode movements through heterogeneous firing rate changes, with many neurons decreasing their activity. In contrast, arkypallidal GPe neurons originate from lateral/caudal ganglionic eminences, express the transcription factor FoxP2, fire at low rates during rest, and encode movements with robust increases in firing. We conclude that developmental diversity positions prototypic and arkypallidal neurons to fulfil distinct roles in behavior via their disparate regulation of GABA release onto different basal ganglia targets.
2021. eLife, 10:e67355