Hippocampal Theta Input to the Amygdala Shapes Feedforward Inhibition to Gate Heterosynaptic Plasticity.
The amygdala and the hippocampus are two brain structures involved in emotional behaviour. Disrupted hippocampus-amygdala interplay appears to occur in anxiety disorders, but the mechanisms of communication between these two brain areas are largely unknown. In this work, we have addressed this issue and discovered that inhibitory nerve cells that use the chemical GABA coordinate such interplay.
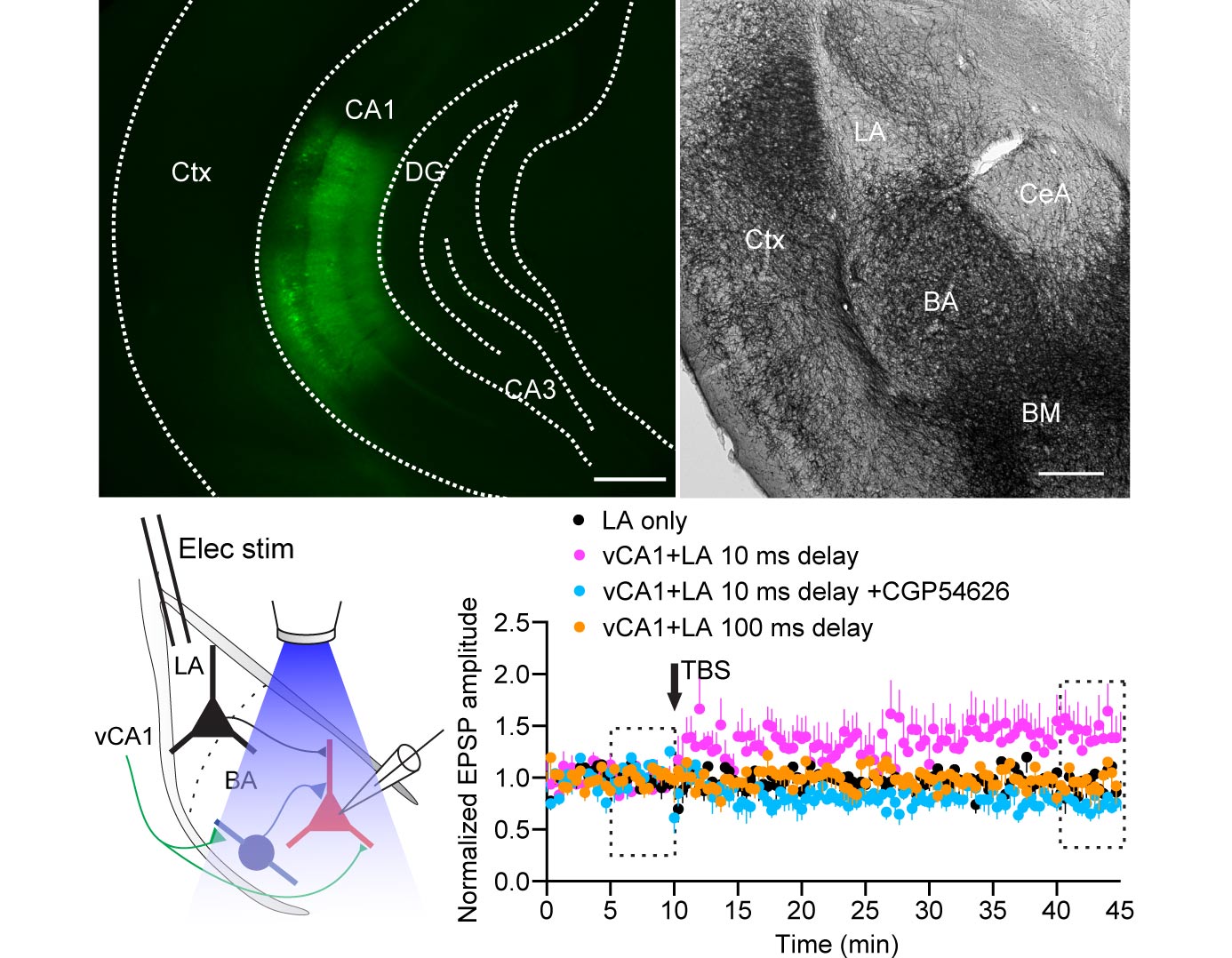
The dynamic interactions between hippocampus and amygdala are critical for emotional memory. Theta synchrony between these structures occurs during fear memory retrieval and may facilitate synaptic plasticity, but the cellular mechanisms are unknown. We report that interneurons of the mouse basal amygdala are activated during theta network activity or optogenetic stimulation of ventral CA1 pyramidal cell axons, whereas principal neurons are inhibited. Interneurons provide feedforward inhibition that transiently hyperpolarizes principal neurons. However, synaptic inhibition attenuates during theta frequency stimulation of ventral CA1 fibers, and this broadens excitatory postsynaptic potentials. These effects are mediated by GABAB receptors and change in the Cl(-) driving force. Pairing theta frequency stimulation of ventral CA1 fibers with coincident stimuli of the lateral amygdala induces long-term potentiation of lateral-basal amygdala excitatory synapses. Hence, feedforward inhibition, known to enforce temporal fidelity of excitatory inputs, dominates hippocampus-amygdala interactions to gate heterosynaptic plasticity. VIDEO ABSTRACT.